Getting Started
The Division of Sponsored Programs (DSP) reviews, approves and facilitates submission of all proposals on behalf of the University of Florida faculty and staff. The PI, department administration support staff and DSP work together as a team to ensure the most competitive and compliant proposals are submitted. All proposals must comply with University, State, Federal and sponsor requirements and PIs must obtain institutional approvals (e.g. Department, College, and DSP approval) prior to submission to the sponsor.
Whether you are new to writing proposals or a seasoned principal investigator, the resources on this website will assist you with:
- Understanding sponsor solicitations
- Developing a proposal in accordance with sponsor guidelines
- Understanding UF-specific proposal requirements (including documentation and eligibility)
- Understanding the process for proposal submission and approval at UF
Learn more about solicitation review and proposal development in course RSH202 via myTraining.
For assistance with general proposal questions, email ufproposals@ufl.edu.
Deadlines
Proposal Submission Deadline Policy
The UF Proposal Submission Deadline Policy is available on General Counsel’s website.
Submit-ready proposals must be submitted to DSP by 9 a.m. the business day prior to the sponsor’s published submission deadline to be considered on-time by DSP.
These two days provide DSP with time to identify and resolve any issues with the proposal that are inconsistent with university policy or sponsor submission requirements. This is important as such issues may result in the inability for the university to carry out the work within the budget submitted or in the proposal being rejected by the sponsor without review. If issues are identified during DSP’s review, the Principal Investigator and relevant unit administrator will be contacted to address issues and facilitate submission to sponsor.
DSP facilitates communication and provision of many other documents to sponsors including pre-proposals and Letters of Intent, Just-in-Time requests, revised budgets, certifications and other information. The expectation is that in order for DSP to have sufficient time to review such information for compliance with University policy and sponsor requirements, we require at least two business days to review. This allows DSP to align the energy required for such a review with its other proposal and agreement obligations.
Examples of Proposal Deadlines:
| Sponsor Due Date | UFIRST Deadline |
| Thursday by 5 p.m. | Preceding Wednesday at 9 a.m. |
| Tuesday by 5 p.m. and preceding Monday is a holiday | Preceding Friday at 9 a.m. |
| Wednesday by 2 p.m. | Preceding Tuesday at 9 a.m. |
| Prime Sponsor indicates Wednesday by 5 p.m., but UF is a subrecipient to an organization with a 3-day internal deadline policy. The lead organizations requires UF to submit documents by 5 p.m. the preceding Thursday to have 1 day to compile and meet their internal deadline. | Wednesday prior to deadline at 9 a.m. |
| Friday, no specific time or time zone mentioned | Preceding Thursday at 9 a.m. |
The UF Proposal Submission Deadline Policy applies to any sponsored program whose solicitation is published by the sponsor at least 30 calendar days prior to the sponsor’s due date.
1. Are solicitations with rolling or continuous open submission windows subject to the internal deadline policy?
The internal deadline applies only to solicitations with specific deadlines from the sponsor. DSP will do everything in its power to submit to a target or rolling deadline, even if provided less than two days to review the proposal. However, DSP will perform a complete review and if there are other proposals received on time for published deadlines, these will take priority.
2. What if UF is a subrecipient to an organization that has its own internal deadline for submission of all proposal materials? (i.e. UF could be a sub to the University of Washington that has a 5 day deadline policy)?
When UF is a subrecipient on a proposal, all materials must be submitted to DSP at 9 a.m. one business day prior to the date that all materials are due from UF to the lead institution.
3. Does the policy apply to “rapid” submissions or short turnaround requirements (i.e. the State needs a response within 10 days)?
Any proposal with less than 30 days turnaround from the date of notice or publication of the solicitation will be exempt from the two day policy.
4. How do I communicate to DSP that a funding opportunity was published less than 30 days before responses are due?
If documented notification of the funding opportunity was made less than 30 days prior to the deadline, the PI or unit contact must add this information to the UFIRST proposal internal documents and make note of it on page 1.0 of the proposal SmartForm.
5. What will DSP do if the proposal is received after the deadline?
Absent of extenuating circumstances, proposals that do not meet the deadline requirements will not be submitted.
6. The policy states that, under extenuating circumstances, late submissions will be accepted. What qualifies as an extenuating circumstance?
Extenuating circumstances include unanticipated closure of the university; serious illness for PI or Co-PI; death of family member; documented sponsor complications or sponsor system technical issues experienced prior to the deadline. In the event of an extenuating circumstance, the PI or unit should contact DSP immediately with their written justification that also includes approval of the unit Research Dean. No waiver of the two day deadline is granted unless it is in writing from a director of DSP.
7. What does submit-ready proposal mean?
“Submit-ready proposal” is defined here.
8. How will DSP handle an oversight by the PI that leads to missing information?
If the review by DSP reveals missing or incomplete materials, DSP will attempt to contact the unit and PI to secure the missing materials and complete submission. Similarly, if an error is discovered in the submission, DSP will attempt to correct the error, or contact the unit and PI for correction of the error.
9. What if the PI or unit discover an error or missing document after submitting to DSP but before the sponsor deadline?
The PI or unit should immediately contact DSP to determine if the correction can be made. If a Core Office Contact has been assigned to the proposal, you may reach out to them directly. If a Core Office Contact has not yet been assigned, contact ufproposals@ufl.edu for assistance.
10. What if an award or agreement arrives in DSP without having been submitted through DSP?
It is University policy that all proposals and communications of proposed work with sponsors be reviewed by the appropriate institutional officials including the Chair, Dean, Director, (or their designees) and DSP prior to submission. Review must occur prior to receipt of agreements and awards to avoid conflicts between funding documents and institutional policies such as F&A rate application and PI eligibility.
A UFIRST proposal must be entered to capture any new funds. DSP will perform the complete review at the time of receipt of the award or agreement. If there are any conflicts, budget discrepancies, or compliance issues identified, they must be resolved (even if this results in the need for additional funds requested from the sponsor or decrease in direct costs available to the project) before DSP will proceed with acceptance of the funds and allowing spending on the project.
If there was a published deadline, DSP will also notify the Chair, Director and Associate Dean to ensure appropriate communications with the PI. Repeat occurrences may result in additional discussions with the PI and the Office of Research.
Required Documents
The proposal must be submit-ready when it arrives to Division of Sponsored Programs for review. The following documents are required at time of proposal review:
- Complete proposal/application package including the scope of work, final budget and budget justification, and sponsor required forms. If the PI is submitting, the complete submission package MUST be loaded into UFIRST for DSP’s review and approval, prior to submission to the Sponsor.
- Proposal submission instructions.
- Sponsor’s solicitation document or proposal guidelines, including program solicitation number if available. Links may expire, so best practice is to upload a PDF copy of the solicitation document. If there is no formal solicitation, include any information regarding submission requirements and/or any communication regarding the program or project.
- If applicable:
- Complete subaward package
- Documentation of sponsor’s published indirect cost policy
- Documentation of any cost sharing requirements
What does submit-ready proposal mean?
A submit-ready proposal is a submission to DSP that is ready for immediate review and submission to the sponsor, with all approvals and permissions provided.
- Disclosure Profiles for key personnel must be up to date in eDisclose ; and
- For all Federal proposals, Research Security Training must be up to date
- UFIRST routing has been completed and the UFIRST proposal record is in a state of Core Office Review including all cost share and department and chair approvals; and
- The proposal is complete and ready for submission to the sponsor indicated by:
- If submitted via UFIRST SF424: all SF424 forms are complete, including all required attachments, and SF424 has been validated to ensure there are no errors preventing submission.
- If submitted through the sponsor’s electronic portal: (1) the PI has given DSP access to submit or certify the proposal in the sponsor system if applicable (e.g., submit access in Research.gov, access in proposalCentral, NSPIRES locked & linked, “Complete and Notify AOR” button clicked in Grants.gov, etc.); (2) the proposal includes all required sponsor forms, components, sections, and attachments, per sponsor program announcement and policy; and (3) a PDF of the complete submission from the portal, or if unavailable, copies of all sponsor forms, components, sections, and attachments, are attached in UFIRST.
- If submitted by email or another method outside of an electronic portal: complete proposal in final format is attached in UFIRST, ready for submission to the sponsor. This includes all required sponsor forms, components, sections, and attachments, per sponsor program announcement and policy.
- If UF is the lead institution on a multi-institutional proposal, all partner institution documentation must be available for review, including required sponsor forms, components, sections, and attachments, per sponsor program announcement and policy.
Proposal Approvals
Delivering Proposals to DSP & Signature Requirements
UF requires approvals for proposals, supplemental funding, progress reports with detailed budgets or other sponsored projects in UFIRST. UFIRST will electronically route the proposal and collect the required approvals.
UFIRST approval workflow requirements will depend on the Department and/or College. The typical workflow of a proposal in UFIRST is a follows:
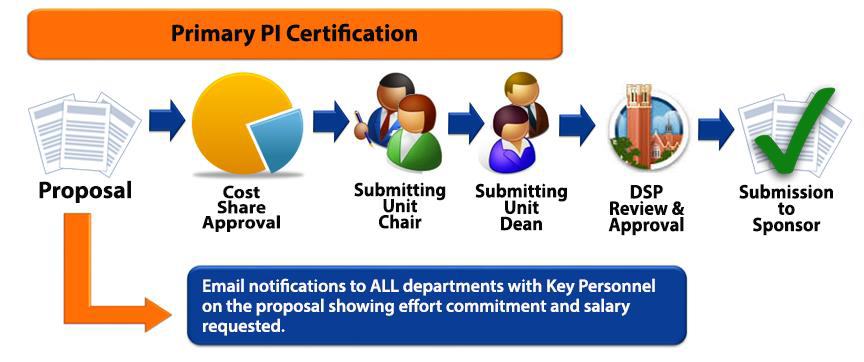
Approvals are to be collected prior to submitting the proposal to a sponsor. In those cases where the approvals were not collected prior to submission, they will be required prior to accepting an award. Approvals are collected using UFIRST.
Proposal Approval Process
- Develop proposal in compliance with University, sponsor, state and federal policies and regulations. The technical description of the work, narratives included in support of that description (e.g., budget justifications) and the ability to execute fall to the PI.
- Identify key personnel and provide reasonable estimate of the effort required to carry out the statement of work.
- Identify resources required to complete the proposal including personnel, facilities, subrecipients, costs to charge the sponsor and resources to be committed by the PI, department, college, or University.
- Identify compliance requirements (i.e. human subjects, animals, safety concerns, export requirements and personal conflicts) required to be managed in performance of the project.
- Complete all required disclosures of Financial Interests and Outside Activities prior to submission of the proposal.
- Identify type of work to be undertaken to facilitate proper selection of facilities and administration (F&A) rate types and category or purpose for institutional reporting.
- Certify to the veracity and integrity of the full and complete proposal including technical and financial commitments.
- Ensure completion by PI and/or study team of all training required for compliance with University, sponsor, state and federal policy and regulations.
- Ensure the proposal is completed with sufficient lead time to allow for the full and complete review by the Chair, Dean, and DSP outlined below.
- Prepare responses to Sponsor inquiries during review process (e.g., Just-In-Time [JIT], revised budgets, revised scope of work, etc.)
In coordination with and support of the PI:
In coordination with and support of the PI:
- Develop a budget with:
- Costing in accordance with University, sponsor, state and federal policy and regulations including only allowable costs.
- Complete and accurate listing of all key personnel and all effort commitments for such personnel and appropriate salaries and fringes are charged for all personnel.
- Correct F&A determination and calculation. If the F&A rate deviates from the federally negotiated rate, PIs with the support of department administration should confirm that the F&A conforms to University policy. Any questions should be sent to DSP in advance of completing the budget and routing the proposal.
- Clear identification of any cost sharing—voluntary or mandatory—provided by University.
- All necessary supporting documentation (e.g., letters of support from representatives authorized to bind the subrecipients, budgets from subrecipients, quotes for equipment).
- Ensure proposal meets sponsor guidelines for items such as page limits, cost limits, and forms required.
- Provide DSP a copy of the proposal guidelines (especially those with special requirements) in advance of the submission deadline for review.
- Contractual exceptions and reps & certs must be handled by DSP and completed prior to submission day.
- Small Business Subcontracting Plans, if applicable, will be coordinated between the Department Administration, DSP and Small Business and Vendor Diversity Relations. Note that completion of these plans takes significant time. Should the proposal require a Small Business Subcontracting Plan, the department administration and PI should notify DSP at least two weeks prior to the proposal deadline.
- If proposal is electronically submitted, facilitate the upload of all proposal documents to appropriate electronic system.
- Complete the appropriate University routing forms in UFIRST.
- Upload all proposal related documents in UFIRST. Pertinent documents include:
- The Solicitation
- Full and complete proposal
- Disclosure of Financial Interests forms, as required
- Subawardee scope, budget and consortium letter, as required
- Facilitate approval through UFIRST.
- Ensure compliance with all College and Department concerns and policies.
- Assist PI with preparation of responses to Sponsor inquiries during review process (e.g., JIT, revised budgets, revised scope of work, etc.). Transmit these responses through UFIRST. DSP will review and coordinate appropriate response to the Sponsor.
- Establish departmental procedures supporting University policies and procedures for reviewing proposal development and routing.
- Provide necessary resources for faculty to complete research under the auspices of the department’s mission.
- Oversee PI and department administration in the preparation and submission of the proposal budget in accordance with University and sponsor policies, and state and federal laws and regulations.
- Certify that any proposal submitted by the department contains all costs necessary to complete the proposal activities and that any costs not identified and committed as cost sharing or simply not included will be borne by the department. Department will bear any errors in calculations resulting in less than sufficient funds.
- Review F&A requested and ensure the rate conforms to the University’s F&A Cost Policy.
- Review and approve the PI eligibility of a non-tenure track faculty member to serve as PI on University submissions, if applicable.
- Review and, if applicable, approve any cost sharing committed in the proposal.
- Establish college/unit procedures supporting University policies and procedures for reviewing proposal development and routing.
- Clearly delineate the college/unit expectations in meeting the department administration responsibilities for proposal development and routing.
- Provide direction, resources, and oversight to ensure that the college administers sponsored programs in accordance with University and sponsor policies, and state and federal laws and regulations.
- Certify that any proposal submitted within the college contains all costs necessary to complete the proposal activities.
- Validate that F&A costs requested conform to the University’s F&A Cost Policy.
- Review and, if applicable, approve any cost sharing committed in the proposal.
- If applicable, review and approve the PI eligibility of a non-tenure track faculty member to serve as PI on University submissions.
- Develop institutional policies and procedures and direct institutional systems and trainings to ensure efficient and effective proposal management practices. In the area of proposal management, this includes but is not limited to, proposing effort and other direct costs, cost sharing, F&A costs, PI eligibility, and proposal signature requirements.
- Provide institutional data systems for managing proposal routing and collecting institutional proposal data. Provide policies, procedures, and training for use of these systems.
- As provided by PI and study team and as time allows, review any proposal solicitation for contractual exceptions, representations and certifications, and other common institutional requirements. Communicate common pitfalls or unusual requirements to the PI and Department Administration.
- As provided by PI and study team and as time allows, perform the final review of the proposal before submission to the sponsor. As time allows, DSP will:
- Confirm the institutional data presented
- Confirm the appropriate F&A rate and any justification for deviations from the federal negotiated rate are included
- Confirm the proper fringe benefit rates are included
- Confirm that the proposal meets the solicitation requirements for page limitations, cost limitations and other proposal limitations
- Act as institutional signatory on all proposal and proposal related documents under Power of Attorney designated by the President of the University.
- Register the University in sponsor electronic research administration systems and provide guidance to campus on account requirements for those systems. Provide overall institutional leadership for electronic research administration planning and implementation.
Resources
- Institutional Profile: Frequently Used Facts
- Principal Investigator Eligibility
- Grants.gov Submission
- Proposal Electronic Systems
- Budgeting Information
- Subawards at Time of Proposal
- Multiple PI Projects (generally for NIH)
- Pre-Proposals, Letters of Intent & White Papers
- RSH202: Solicitation Review & Proposal Development
Last updated 7/14/25
